Chemical Dominoes
This lab's purpose was to use recently learned knowledge in chemistry to create a series of reactions to light up a light bulb. In the video below is our domino thing and as you'll see our reactions with the chemicals worked fine, but we had more difficulty with the actual dominoes that we had set up. If they'd cooperated, we'd have had a perfect run.
The science behind our chemical components are as follows:
Yellow Balloon in beginning:
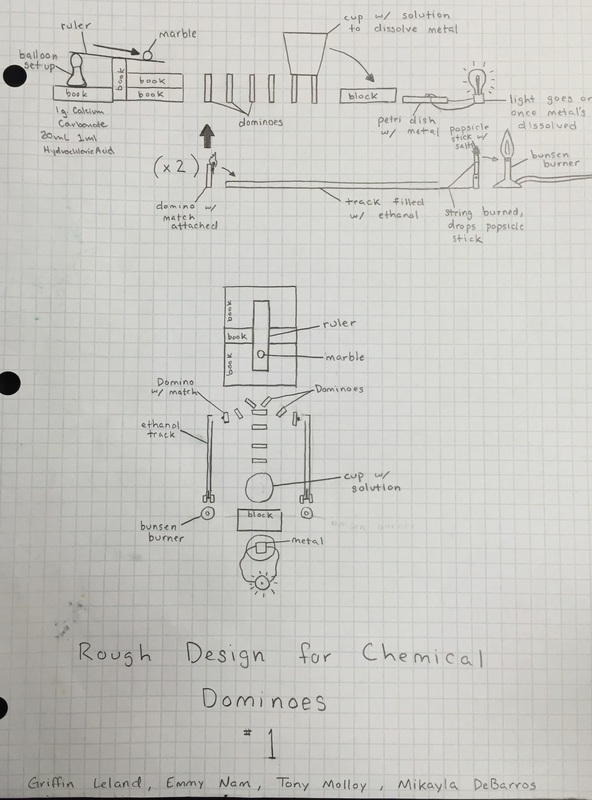
In the glass beaker we combined 10mL of hydrochloric acid with 2 g of calcium chloride. When combined, these substances create carbon dioxide which filled the balloon which in turn raised the lever, releasing the marble.
Burning String:
To get the string to burn effectively we soaked the part of it we wanted to burn in alcohol. When heat is applied to alcohol and air a combustion reaction occurs, resulting in a flame.
Colored Flame:
The green flame you see once the popsicle stick drops is the result of burning copper chloride, which produces a unique flame due to copper's atom structure.
Light Bulb Reaction:
To get the light bulb to light up we set up a circuit where a strip of aluminum stopped the bulb from lighting. To break the aluminum we used liquid copper chloride which causes a single replacement reaction to occur. The result is that wherever the copper chloride hit the aluminum, the aluminum metal would turn into liquid aluminum chloride and leave very small traces of copper behind.
Below is an early blueprint of our design
Reflection
Our project worked half as well as we wanted and their were many things we could've improved. One thing we could've spent more of our time on was getting the dominoes to fall the way we wanted to, it was consistently the biggest obstacle for us and we didn't focus on it enough. Another obstacle that was on and off was the accuracy of the styrofoam cup pouring the liquid we needed onto the aluminum, this was a constant battle of chance for us and if given more time we would've been able to solve it. At the same time as we were trying to overcome these challenges, we found a lot of success. We got the string and popsicle stick to burn/fall the way we wanted to on our first try, pretty much, and I think our group had really nice cooperation. We all worked on something at any time and were usually working on seperate parts of the reaction in order to bring it all together.
Our project worked half as well as we wanted and their were many things we could've improved. One thing we could've spent more of our time on was getting the dominoes to fall the way we wanted to, it was consistently the biggest obstacle for us and we didn't focus on it enough. Another obstacle that was on and off was the accuracy of the styrofoam cup pouring the liquid we needed onto the aluminum, this was a constant battle of chance for us and if given more time we would've been able to solve it. At the same time as we were trying to overcome these challenges, we found a lot of success. We got the string and popsicle stick to burn/fall the way we wanted to on our first try, pretty much, and I think our group had really nice cooperation. We all worked on something at any time and were usually working on seperate parts of the reaction in order to bring it all together.